It’s no secret that getting RWE (Real World Evidence) research done in the EU has historically been challenging. This is in contrast to the US where data including EHR and claims data is made available relatively seamlessly from multiple options of pooled data sets from many health systems that can be licensed for research purposes. One common solution for getting research done in the EU has been to partner with health systems to conduct research at each institution through processes similar to a clinical trial using sponsored research agreements. At Graticule we have continued to build out our network of European sites and have handled the requirements from sites working through the necessary processes for project approval, ethics approval, and data access.

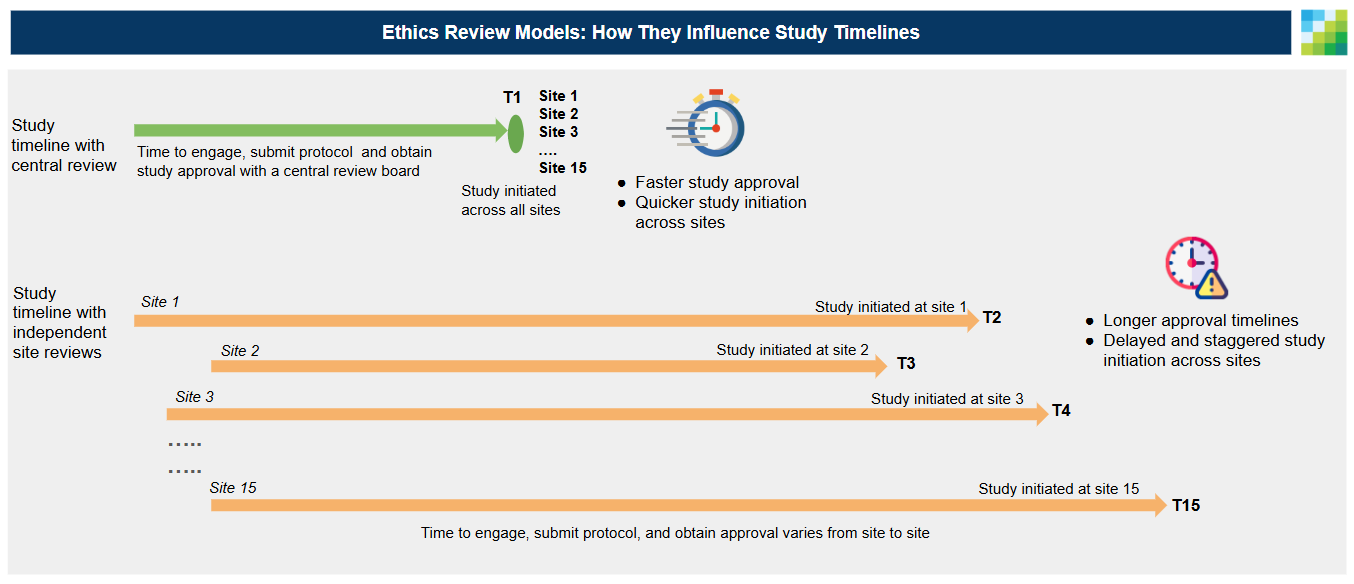
I wanted to point out in this blog that the EU regulatory groups in countries have been looking to improve research efficiency and those steps have been taking effect. In the past year we have seen successful implementation of improved centralized ethics review for Real World Data in Europe with a positive impact on sponsors to get projects up and running quickly. This is making research that may not have been feasible in prior years now possible because site start-up can be streamlined across multiple sites.
The ethics review is a critical path requirement for getting to a project green light. The ethics review step in research on human subjects or medical records in Europe, often called the REB (Research Ethics Board) is similar to the IRB (Institutional Review Board) in the US. The ethics review is a process where a set of information is collected including the protocol and the plans for how the study will be conducted such as how data will be collected, managed, and analyzed. A group of appointed clinicians and administrators from the institution review the combined information to determine if the research meets the ethics standards of the organization. If approved then projects can proceed on the basis of following the stated protocol and approach described in submitted documents. This is an important process but also one that creates issues regarding the speed, cost, and scale of research.
With many small institutions each with their own ethics review processes it is a time consuming and redundant process with potential inconsistency on opinions regarding details in the plan. Keep in mind that protocols and studies are living documents so not only does the initial approval need to go through a review but so do any protocol amendments or renewals of the protocol on a periodic basis for an ongoing study. So replicating this process across 5 or potentially 50 organizations is no small task and could take longer than the total time allocated to run the study.
In recognizing this challenge the United States has had systems since the early 2000s for centralized IRB where the review process can occur in a single central review and then the approved protocol review can be syndicated to multiple sites.
For example, at Graticule we have worked with the company WCG which provides a service for central ethics oversight of research protocols. This central approach operates in both real world evidence programs and in clinical trials. It was enabled in 2006 through guidance from the FDA (Using a Centralized IRB Review Process in Multicenter Clinical Trials). We have found in working with health systems in the US that central IRBs greatly streamline the process for ethics review. We often still submit the same information to the site ethics review group at each site but having an outside IRB approval along with the materials is sufficient for driving the process on an expedited basis of weeks vs. months at sites potentially with limited to no additional review.
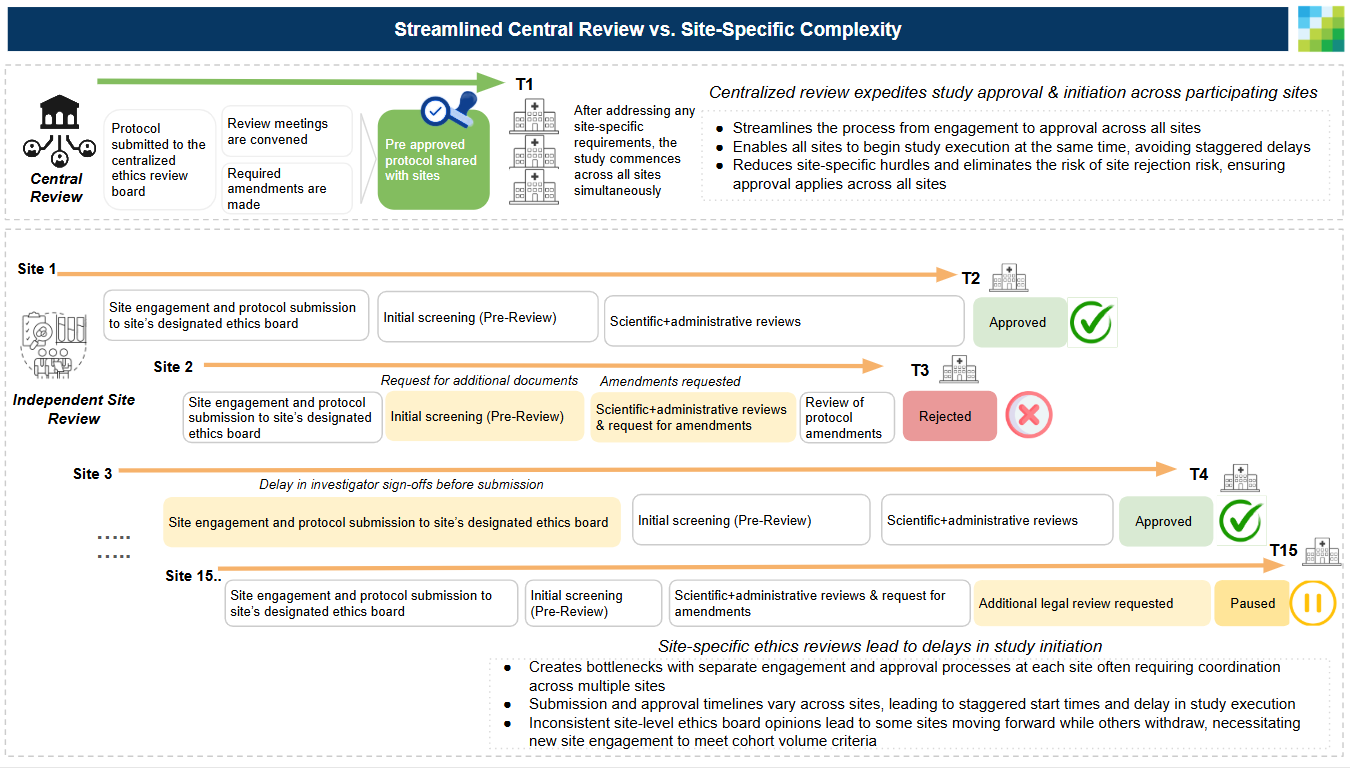
Central ethics review for Real World Data at EU health systems wasn’t the case when Graticule started working on EU projects in 2019. Each study we proposed at a site had to go through the process of full ethics submission and approval at the local institution on or before contracting to add the site to a research network for a study. Clinical trials in Europe have had no choice but to encounter a daunting process of going through an institutional review board at each site or local region given the challenges of recruitment of sufficient patients. This site-specific approach introduces considerable challenges and complexities as each site follows its own submission and approval process. The result is inconsistent timelines, increased administrative burden and an increased risk of site withdrawal. This fragmented process also leads to variability in approval decisions across site specific ethics boards, slowing down study timelines and complicating large-scale research coordination.
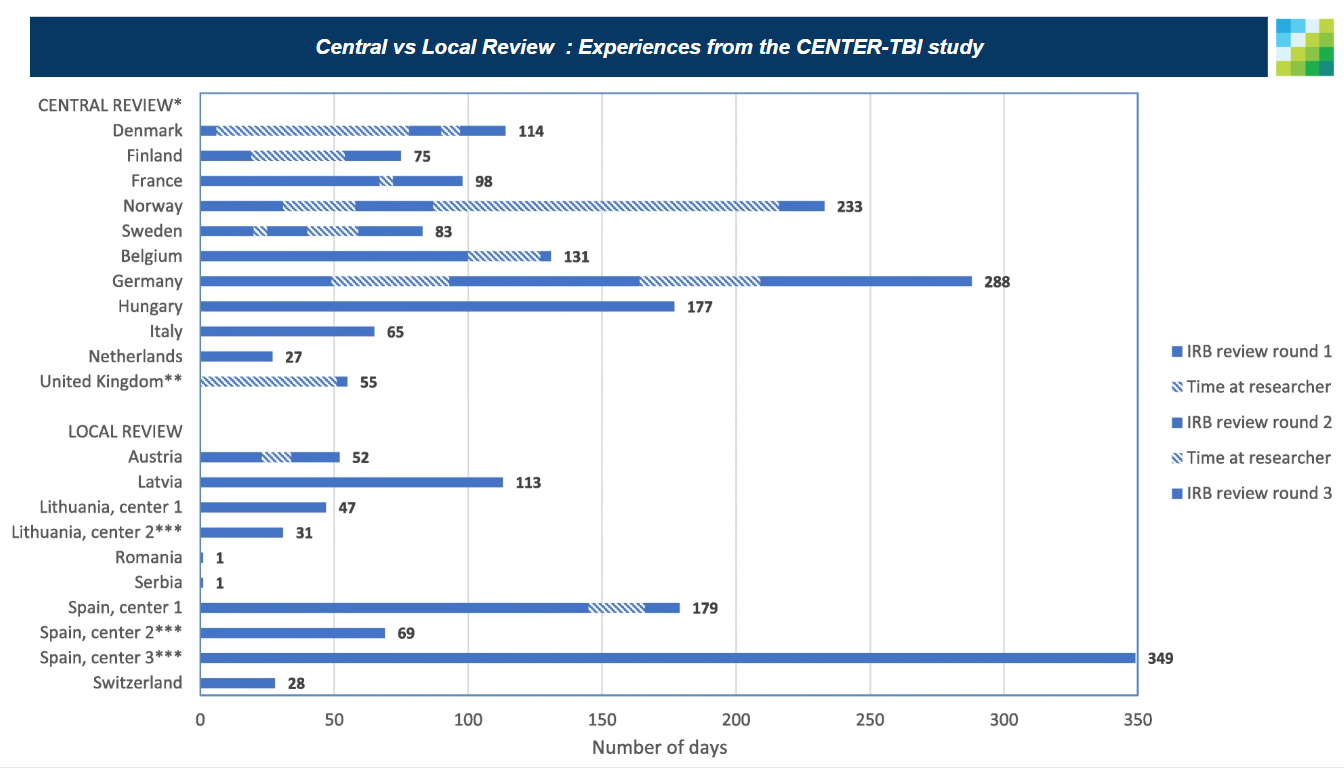
Ethics review is a time consuming process. For reference, look at the time to complete institutional review by country for a prospective observational study in 2020 in the article: How do 66 European institutional review boards approve one protocol for an international prospective observational study on traumatic brain injury? Experiences from the CENTER-TBI study. The article showed that ethics review can take hundreds of days to complete at individual sites. But also it showed that central review allowed multiple sites to effectively share an ethics review efficiently.
Graticule has been working in Italy on a number of real world data projects for sponsors. In Italy, the move toward a single Ethics Committee approval for multi-center studies was initiated by Legislative Decree No. 52/2019, aimed at implementing EU Regulation 536/2014. While introduced in 2019 it was initially only applicable to clinical trials. The Italian legislation was gradually extended to also apply to non-interventional observational studies. By mid-2024, most multi-center observational studies in Italy have now transitioned to one coordinating Ethics Committee providing a single opinion, with local sites typically only requiring feasibility or administrative clearance rather than separate ethics approvals.
2024 was the first year for us to experience the changes in the ethics review process in Italy. Keep in mind that while in the US we work with a private commercial organization for central ethics review in Italy the ethics review is a public function. In Italy they have instituted regional levels organizing their ethics review. Italy is made up of 20 regions. For example Tuscany and Sicily are regions. In practice from the new legislation the central ethics review allows sponsors to work through one regional research ethics board (REC) such as Tuscany and then use the guidance from that region to be used in local ethics boards across multiple regions such as Sicily including the many health systems in that region. In our experience 2024 was a year of working through the process for the first time for RWD protocols. In 2025 the scale-up of the process is beginning to work and if systems can become streamlined this can significantly reduce the complexity of conducting Real World Data projects in Italy. With frameworks already adopted at many health systems for research aligning data sets to OMOP through OHDSI we see a scalable solution to combine ethics review with data harmonization going forward in Italy.

We provide Italy as an example where we have seen good progress on central REC recognition in Real World Data but similar efforts are underway across the EU-4 countries (Italy, Spain, France, and Germany) that are a priority in life science research. From a broader perspective the European Health Data Space provides additional new emphasis for enabling research data access. In January 2025 the new regulation on the EU HDA that was approved in April 2024 was posted and is available for any regulatory team to review to consider the potential impact of the EHDS. The EHDS regulation was a response to how necessary it was to act quickly during COVID to collect and analyze data to meet a health crisis. It includes the following statement emphasizing the importance of Real World Data.
“Electronic health data used for secondary use can bring great societal benefits. The uptake of real-world data and real-world evidence, including patient-reported outcomes, for evidence-based regulatory and policy purposes as well as for research, health technology assessment and clinical objectives should be encouraged. Real-world data and real-world evidence have the potential to complement health data currently made available. To achieve that goal, it is important that datasets made available for secondary use pursuant to this Regulation be as complete as possible. This Regulation provides the necessary safeguards to mitigate certain risks involved in the achievement of those benefits. The secondary use of electronic health data is based on pseudonymised or anonymised data, in order to preclude the identification of the data subjects.”
More summary information is available directly from the EU health regulatory web site but a key concept that is outlined is to provide “data permits” for uses that are ethical in research. The structure of these data permits will allow organizations to access the centralized research repositories being built in Europe. Programs such as H2O, an IHI project around patient reported outcomes have been established to support research networks across geographies. It has built commitment from both health systems and life science companies working together to build a path to data sharing. In Germany, for example, a number of large health system sites pooled patient data into what is available through the German Portal for Medical Research. A large volume of deidentified patient data has been aggregated for Real World Data use. While it is still quite early in the development of the data repository including the governance and model for life sciences collaboration it is a strong opportunity to now work through the end to end lifecycle of RWD projects.
In short the EU continues to move forward with increasing the efficiency of initiating RWD research and executing it at larger scales. We believe that it is still important to factor that timelines will be more drawn out in Europe. So it is still advised to start building long term relationships early and also work with a team like Graticule who has invested in working within the landscape of sites and building relationships and expertise that can be extended to new studies. We look forward to the new work in 2025 forward and expect based on the status of these changes that the regulatory environment in the EU will continue to drive increasing efficiency and scale through new governance approaches and collaborations between institutions.
