eSourcing patient data from Electronic Health Records can revolutionize long-term patient tracking for new and emerging treatments
Cell and gene therapies (CGT) are designed to deliver long-lasting, often permanent effects on the human body, but they carry risks of long-term side effects like autoimmune disorders or certain cancers. In response, regulations—including FDA guidelines published in 2020 and similar mandates across Europe—require extended periods of patient monitoring, sometimes as long as 15 years.(1) Known as long-term follow-up (LTFU), these monitoring requirements pose significant challenges.
Over a decade or more after CGT treatment, patients may switch providers, be lost to follow-up, or pass away. Patients slip beyond the reach of a particular healthcare system’s records, leaving blind spots in the record of the patient’s journey. Further, patient tracking often requires that the patient take an action such as complete a survey, fill out a form, or attend a special appointment outside of their normal course of care. Naturally, relying on and keeping patients engaged in these activities can be quite difficult.
On the data collection side, monitoring is labor-intensive and costly. Often, the research coordinators responsible for patient monitoring will gather data from multiple sources and manually transcribe the information. This is a lot of work, and these busy, trained professionals are typically responsible for many non-administrative tasks. Most smaller community hospitals lack these staff resources and the overall infrastructure to handle the effort required. As a result, CGT companies only end up collaborating with a limited number of sites with the capacity to support LTFU.

Current approaches to long-term follow up and data collection are expensive and limiting. They require a lot of effort, and often rely on active patient engagement. How can we streamline processes to ensure efficient, cost-effective monitoring over long periods? eSourcing—automated EHR data transfer into CGT research studies—can be the game changer the industry needs. When health systems and sponsors connect, EHR data flows continuously, passively in the background. Staff at health systems is then free from the burden of manual entry of data.
Health Information Exchanges for Gene and Cell Therapy: a big opportunity
I’ve blogged about the benefits of Health Information Exchanges (HIEs) for research before, and will revisit it here as they are very relevant in this space. eSourcing through HIEs can establish a broad, national surveillance network for CGT patients. Unlike a single-sponsor to individual-health system approach to research data, use of HIEs provides a way access consented data from broad patient populations. The National Institute of Health’s “All of Us” pilot program is a great example. We see a lot of potential in this approach; our eSourcing framework is currently being adapted to establish connectivity with HIEs to support efforts such as CGT.
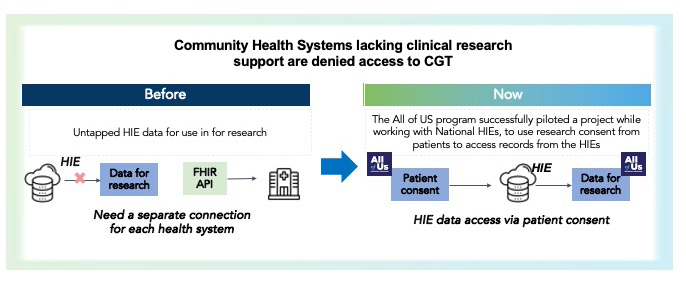
Whole population data access possible through HIE’s solves one piece of the puzzle to understand the impact of CGT on patient safety and outcomes. Another puzzle piece involves the quality and completeness of the data itself. Depth, beyond just breadth. We’ve been working on this as well.
Solving for data “missingness”
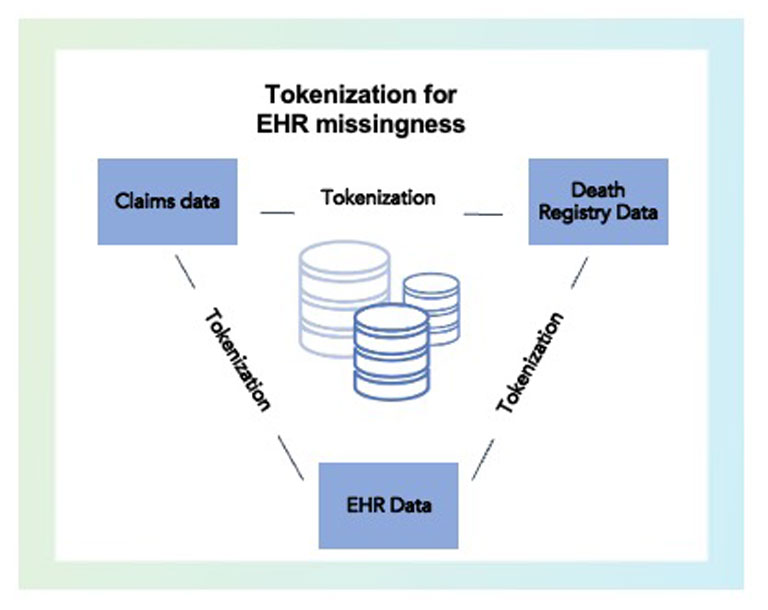
Gaps in EHR data can occur when patients visit multiple providers and health systems, as they likely would over a period of many years. These lead to blind spots in a patient’s medical history. Closed claim data from payers, insurers and providers can help fill these gaps and provide more complete view of the patient’s journey. We use tokenization to link licensed claims and even death registry data, all while ensuring we maintain a compliant and secure health profile. Ultimately, the research clinicians get a better picture of what is going on with the patient.
A multi-pronged approach to LTFU
Effective LTU requires more than just the technology (eSourcing, tokenization)—it requires a consent framework designed to accommodate current and future data needs. As the CGT landscape evolves, you can future-proof your programs by implementing strategies that align with current practices while anticipating future advancements. This ensures your data collection systems remain adaptable and ready for what’s next.
Contact our team to get started> Graticule has extensive experience in the effective use of EHR data for research. If you want to tackle the issue around LTFU and boost your bottom line, reach out. We can share success stories, plan pilot projects, and guide you toward early wins with tangible results.
(1 )Nat Med 27, 563 (2021). https://doi.org/10.1038/s41591-021-01333-6